 » Barrier-free View
» Barrier-free View
| ||||||||
 | ||||||||
 |
 |
 |
| Picture of the 1st Network Meeting of the Herz-Kreislauf-NetzDownloadsArchivExternal job opportunities |
 |
Zebra fish mutants as a model for human cardiomyopathies
 |
Subproject leader
Dr. Wolfgang Rottbauer
University of Heidelberg
Internal Medicine III / Cardiology
Otto-Meyerhof-Center
Im Neuenheimer Feld 350
69120 Heidelberg
email: wolfgang.rottbauer@med.uni-heidelberg.de
Phone: 06221-56 4697
The identification of the main genetic causes and molecular pathways underlying human dilated cardiomyopathies (DCM) has been hindered by their significant mortality and the low percentage of familial forms, suitable for classical linkage studies. Hence, for the systematic dissection of the genetic pathways involved in the pathogenesis of this disease entity and the development of better therapeutic strategies, genetic animal models like the zebrafish (ZF) are required. In two large-scale ZF mutagenesis screens, more than 80 mutant lines with impaired cardiac function have been identified. We (funded by NGFN-1) and others recently isolated in some of these lines novel genes essential for the development of proper vertebrate heart form and function (Fig. 1). Several of the identified genes have also been found to be mutated in human DCM making the characterized ZF mutants to disease models for human DCM.
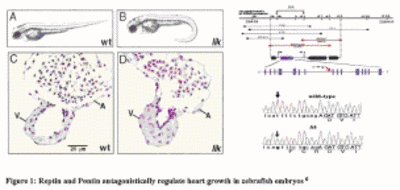 |
Fig1: Reptin and Pontin antagonistically regulate heart growth in zebrafisch embryos. |
To facilitate the identification of the main genetic causes of human DCM and to characterize the underlying molecular pathways, the proposed research now aims to characterize 20 novel recessive, embryonic-lethal mutations in ZF, which exhibit reduced or absent contractility of the ventricular and/or atrial heart chambers (Fig. 2).
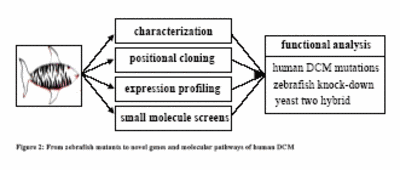 |
Fig.2 From zebrafih mutants to novel genes and molecular pathways of human DCM. |
During the course of this study 20 novel embryonic-lethal, recessive mutant zebrafish lines with reduced cardiac contractility were bred, outcrossed and the cardiac phenotypes structurally, molecularly and functionally characterized. Currently the mutant genes are islolated by positional cloning and timing and location of mutant gene expression at RNA and protein levels is determined. Loss-of-function and cardiac-specific gain-of-function studies using cardiac-specific ZF promoter constructs are performed with the corresponding genes. In collaboration with D. Weichenhan, Heidelberg, (KG-CV3.1) we determine by mutation screening in large patient cohorts - the role of the identified genes in human DCM and cardiac malformations. In cooperation with RZPD-Berlin and MPI Tübingen we already determined cardiac-specific gene expression profiles of 8 ZF mutant lines which were identified in NGFN-1. The expression pattern and basic function of the differentially regulated genes is currently characterized using RNA in situ hybridisation, gene knockdown (GeneTools, USA) and cardiac-specific overexpression strategies in ZF (cooperation with KGCV-net project 3). In cooperation with N. Frey/S. Wiemann, Heidelberg (KG-CV3.4 and SMP-Cell) we are in the process of overexpressing the novel mutant genes in mammalian cell lines to evaluate their sub-cellular localization and impact on basic cell functions (e.g. cell growth, mitosis, apoptosis). Novel interacting protein partners are currently identified in two-hybrid screens. Soon, we will start to test the impact of small molecule libraries on the ZF mutant heart phenotypes using high throughput screening methods. These studies will be performed in collaboration with M. Fishman/F. Serluca (Novartis, Boston, USA) and R. Peterson and C. MacRae (MGH, Boston, USA).
Using the outlined approach, we recently identified the genetic defect in zebrafish dead beat and were able to show for the first time that Vascular Endothelial Growth Factor (VEGF) signalling modulates cardiac contractility through its receptor FLT-1 and consecutively activation of Phospholpase C g1 (Figure 3).
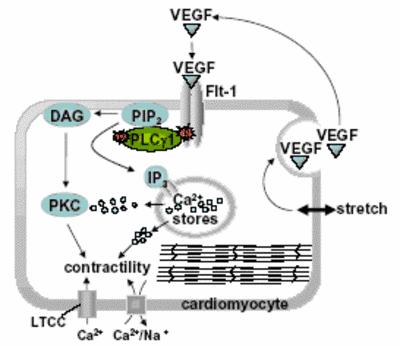 |
Fig.3 VEGF - A novel regulator of cardiomcyte contractility |
The proposed research (see Fig. 2) is expected to reveal new causal genes and genetic pathways for human DCM and, eventually, will support the design of new treatment regimens.
Publications:
1. Rottbauer, W., Grünig, E., Brown, B.D., Zehelein, J., and Scheffold, T. 1996. A novel ß-myosin heavy chain mutation in a large family suffering from dilated and hypertrophic cardiomyopathy. Circulation 94, 8, Suppl I: 162-163. 94:162-163.
2. Rottbauer, W., Gautel, M., Zehelein, J., Labeit, S., Franz, W.M., Fischer, C., Vollrath, B., Mall, G., Dietz, R., Kubler, W., et al. 1997. Novel splice donor site mutation in the cardiac myosin-binding protein- C gene in familial hypertrophic cardiomyopathy. Characterization Of cardiac transcript and protein. J Clin Invest 100:475-482.
3. Grunig, E., Tasman, J.A., Kucherer, H., Franz, W., Kubler, W., and Katus, H.A. 1998. Frequency and phenotypes of familial dilated cardiomyopathy. J Am Coll Cardiol 31:186-194.
4. Stainier, D.Y., Fouquet, B., Chen, J.N., Warren, K.S., Weinstein, B.M., Meiler, S.E., Mohideen, M.A., Neuhauss, S.C., Solnica-Krezel, L., Schier, A.F., Fishman M.C. 1996. Mutations affecting the formation and function of the cardiovascular system in the zebrafish embryo. Development 123:285-292.
5. Rottbauer, W., Baker, K., Wo, Z.G., Mohideen, M.A., Cantiello, H.F., and Fishman, M.C. 2001. Growth and function of the embryonic heart depend upon the cardiac- specific L-type calcium channel alpha1 subunit. Dev Cell 1:265-275.
6. Rottbauer, W., Saurin, A.J., Lickert, H., Shen, X., Burns, C.G., Wo, Z.G., Kemler, R., Kingston, R.,Wu, C., and Fishman, M. 2002. Reptin and pontin antagonistically regulate heart growth in zebrafish embryos. Cell 111:661-672.
7. Xu, X., Meiler, S.E., Zhong, T.P., Mohideen, M., Crossley, D.A., Burggren, W.W., and Fishman, M.C. 2002.
Cardiomyopathy in zebrafish due to mutation in an alternatively spliced exon of titin. Nat Genet 30:205-209.
8. Sehnert, A.J., and Stainier, D.Y. 2002. A window to the heart: can zebrafish mutants help us understand heart disease in humans? Trends Genet 18:491-494.
9. Gerull, B., Gramlich, M., Atherton, J., McNabb, M., Trombitas, K., Sasse-Klaassen, S., Seidman, J.G., Seidman, C., Granzier, H., Labeit, S., Thierfelder, L. 2002. Mutations of TTN, encoding the giant muscle filament titin, cause familial dilated cardiomyopathy. Nat Genet 30:201-204.
10. MacRae, C.A., and Peterson, R.T. 2003. Zebrafish-based small molecule discovery. Chem Biol 10:901-908.
11. Rottbauer W, Just S, Wessels G, Trano N, Most P, Katus HA, Fishman MC (2005). VEGF-PLCg pathway controls cardiac contractility in the embryonic heart. Genes Dev 19: 1624-163
more information about the working group:
http://www.klinikum.uni-heidelberg.de/index.php?id=4580